There’s a point in chemistry where numbers stop being abstract and start feeling like a way to explain what atoms are actually doing. Formal charge sits right in that space. If you’ve ever drawn a structure and wondered why one version feels more “correct” than another, this idea usually explains it.
People often jump straight into formulas, but it makes more sense when you see how it fits into the bigger picture of bonding and electron sharing. Even tools like a bmi calculator don’t mean much until you understand what the numbers represent, and formal charge works the same way.
How to Calculate Formal Charge?
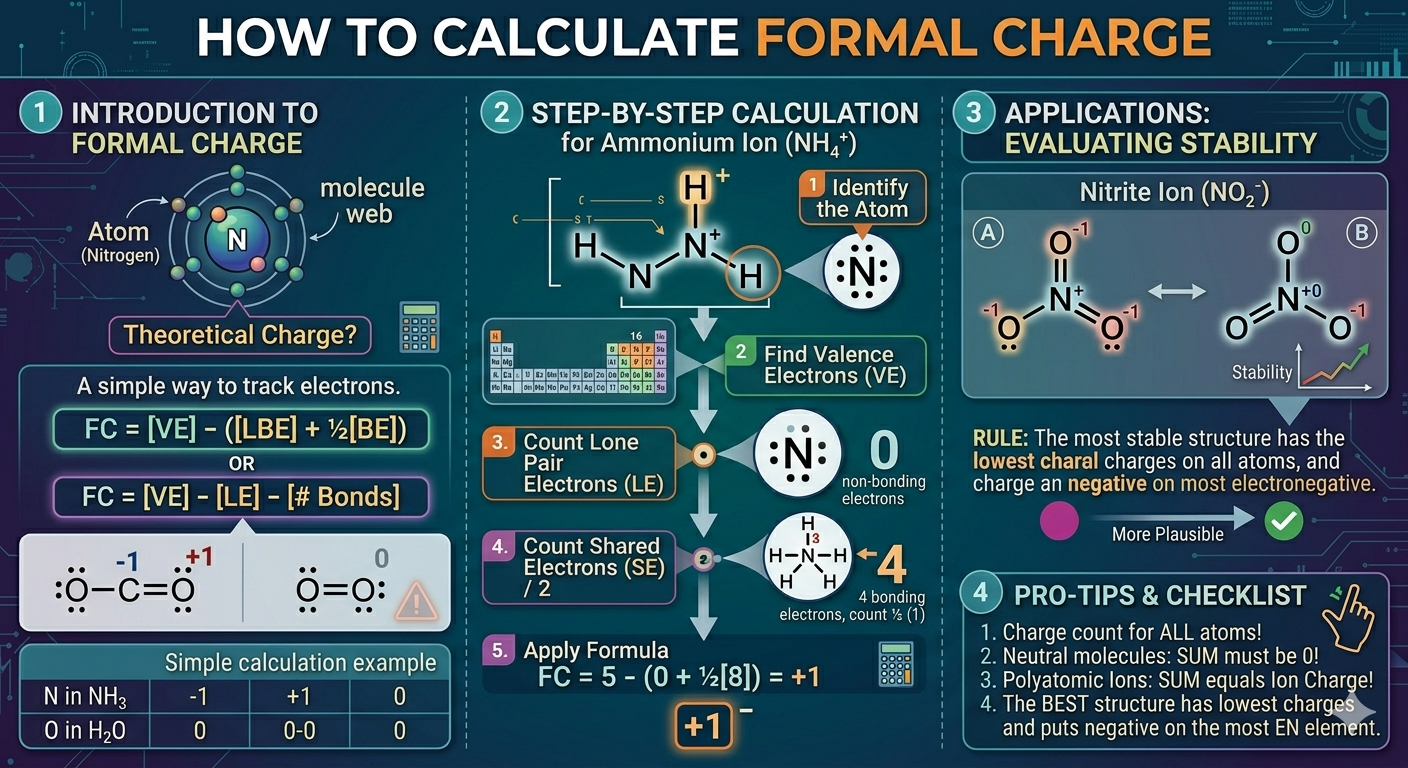
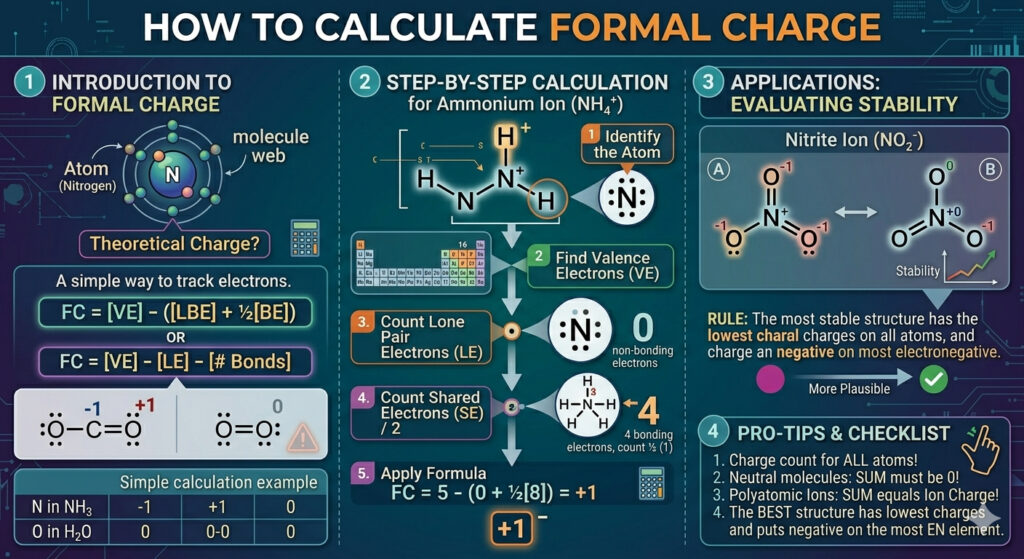
The phrase how to calculate formal charge sounds a bit mechanical, but it’s not just about plugging numbers into a formula. It’s about tracking electrons and seeing how they’re distributed in a molecule. At its core, formal charge compares the electrons an atom “owns” in a molecule to the number it normally has on its own.
The basic formal charge formula looks like this:
Formal Charge = Valence Electrons − Nonbonding Electrons − (Bonding Electrons ÷ 2)
It might seem like a lot at first glance, but each part has a simple meaning. Valence electrons are what the atom brings to the table. Nonbonding electrons are the lone pairs sitting on that atom. Bonding electrons are shared between atoms, so you split them in half. That’s really it. Once you get used to counting, the process becomes almost automatic.
Breaking down the formal charge formula in plain terms
If you try to memorize the formal charge formula without understanding it, it gets confusing quickly. A better way is to think about ownership. Imagine every electron in a bond is shared equally. So if two atoms share a pair, each gets one.
Valence electrons come from the periodic table. Oxygen has 6, nitrogen has 5, carbon has 4. These numbers don’t change. What changes is how those electrons are arranged in a Lewis structure.
Let’s say an atom has:
- 6 valence electrons
- 2 lone pair electrons
- 4 bonding electrons
Then the formal charge would be:
6 − 2 − (4 ÷ 2) = 6 − 2 − 2 = +2
That +2 tells you the atom has “lost” electron density compared to its neutral state.
If you ever feel unsure, using a percentage calculator mindset helps just break things into parts and handle each piece separately.
Why formal charge actually matters?
It’s tempting to treat formal charge as just another chemistry calculation, but it plays a bigger role. It helps you decide which molecular structure is more stable.
When you draw multiple Lewis structures, they’re not all equally good. The best one usually:
- Keeps formal charges as close to zero as possible
- Places negative charges on more electronegative atoms
- Avoids large charge separations
So formal charge becomes a kind of filter. It helps you sort through possibilities and pick the one that makes the most sense chemically.
This is especially important in reaction mechanisms, where small changes in electron distribution can shift how a reaction behaves.
Step-by-step method that actually works
If you’re looking for how to calculate formal charge step by step with examples, here’s a simple flow that doesn’t feel too rigid:
Start by drawing the Lewis structure clearly. Count all valence electrons and place bonds first. Then fill in lone pairs.
Next, focus on one atom at a time. Don’t try to do the whole molecule at once.
For each atom:
- Note its valence electrons
- Count lone pair electrons
- Count bonding electrons
- Apply the formula
Move slowly at first. Speed comes later.
Working through a real example
Take water (H₂O). Oxygen is the central atom.
Oxygen has 6 valence electrons. In the structure:
- It has 4 nonbonding electrons (two lone pairs)
- It shares 4 bonding electrons (two bonds with hydrogen)
So:
6 − 4 − (4 ÷ 2) = 6 − 4 − 2 = 0
Hydrogen:
- Valence electrons = 1
- No lone pairs
- 2 bonding electrons
So:
1 − 0 − (2 ÷ 2) = 1 − 1 = 0
Everything comes out neutral, which matches what we expect.
You can repeat this process for more complex molecules, though it takes a bit more patience.
Using formal charge in larger molecules
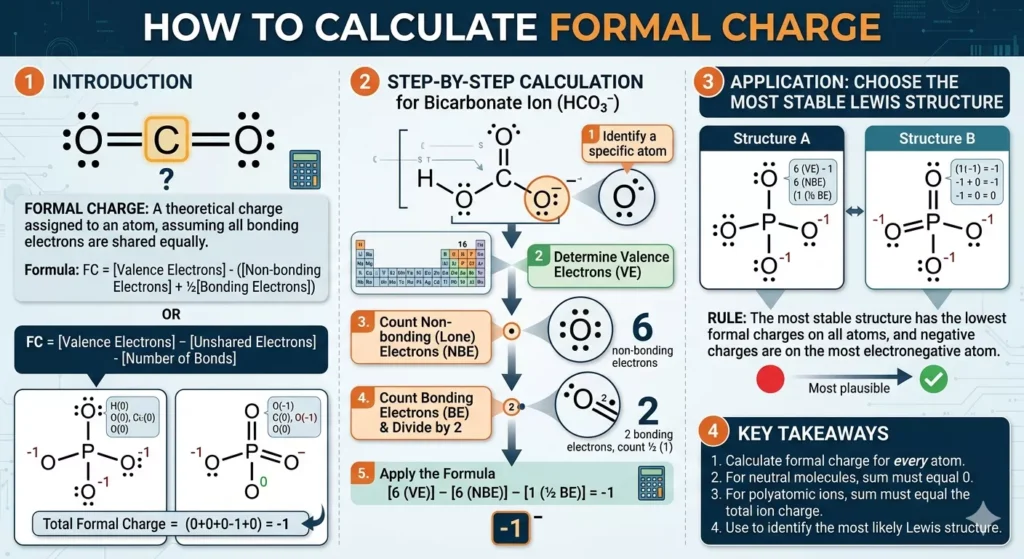
When molecules get bigger, formal charge becomes more useful, not less. You’ll often see multiple possible Lewis structures. This is where formal charge helps narrow things down.
For example, in nitrate (NO₃⁻), different structures shift where double bonds appear. Formal charge helps decide which arrangement is more reasonable.
The pattern you’ll notice:
- Structures with minimal charges are preferred
- Negative charges tend to sit on oxygen rather than nitrogen
This isn’t a strict rule, but it works most of the time.
Some students try to rush through these calculations, but slowing down often saves time later. It’s a bit like using a timezone converter you double-check to avoid mistakes.
Formal charge vs Real charge
There’s often confusion between formal charge and actual charge. They’re not the same.
Formal charge is a bookkeeping tool. It assumes electrons are shared equally. Real charge, or partial charge, comes from actual electron distribution, which can be uneven.
So an atom might have a formal charge of zero but still carry a slight positive or negative character in reality.
That’s why formal charge is useful, but not the full story.
Common mistakes people make
A few patterns show up again and again:
- Forgetting to divide bonding electrons by 2. This is probably the most common error.
- Mixing up valence electrons with total electrons. Always go back to the periodic table.
- Ignoring lone pairs. These are easy to miss, especially in crowded structures.
- Rushing through calculations. Even small errors can throw off the whole structure.
- It’s not unusual to make these mistakes early on. They tend to fade with practice.
Table: Quick reference for common atoms
| Atom | Valence Electrons | Typical Bonds | Common Formal Charge |
|---|---|---|---|
| Hydrogen | 1 | 1 | 0 |
| Carbon | 4 | 4 | 0 |
| Nitrogen | 5 | 3 | 0 |
| Oxygen | 6 | 2 | 0 |
| Chlorine | 7 | 1 | 0 |
This table isn’t strict, but it gives a rough idea of what to expect.
How formal charge connects to Lewis structure
You can’t really separate formal charge from Lewis structure. One depends on the other.
When you draw a structure, you’re making assumptions about bonding. Formal charge tests those assumptions. If something feels off, checking formal charges usually reveals why.
Sometimes you’ll redraw a structure several times before it feels right. That’s normal. Even tools like an age calculator only work when inputs are correct. Same idea here structure first, calculation second.
Applying formal charge in reactions
In reaction mechanisms, formal charge becomes more than just a number. It helps explain movement of electrons.
If an atom carries a positive formal charge, it might attract electrons. If it’s negative, it might donate.
This helps predict how molecules interact.
You start to see patterns:
- Electron-rich areas attack electron-poor areas
- Charges shift as bonds break and form
It’s not always perfect, but it’s a good guide.
Understanding electron distribution better
Formal charge ties closely to electron distribution. It shows how electrons are spread across a molecule.
When distribution is uneven, charges appear. This affects stability and reactivity. Some molecules tolerate charge separation better than others. Oxygen, for example, handles negative charge more comfortably than carbon.
This kind of detail matters more as you go deeper into chemistry. You might even relate it to something like an energy converter, where balance and transfer matter.
Difference between formal charge and oxidation state
- Another common confusion is between formal charge and oxidation state.
- Formal charge assumes equal sharing of electrons. Oxidation state assumes complete transfer.
- So oxidation states are often larger in magnitude.
- Both have their uses, but they answer different questions.
When formal charge doesn’t tell the whole story
There are cases where formal charge alone isn’t enough. Resonance structures, for example, spread charge across multiple atoms.
In those cases, no single structure is perfect. Instead, the real molecule is a blend. Formal charge still helps, but it’s part of a bigger picture.
Tips that actually help in exams
If you’re preparing for exams, a few habits make a difference:
- Write out the formula every time, even if you think you remember it.
- Double-check electron counts before calculating.
- Keep structures neat. Messy drawings lead to mistakes.
- Practice with different molecules. Repetition builds speed.
- It’s less about memorizing and more about getting comfortable with the process.
Table: Example formal charge calculation
| Molecule | Atom | Valence e⁻ | Lone e⁻ | Bonding e⁻ | Formal Charge |
|---|---|---|---|---|---|
| NH₃ | N | 5 | 2 | 6 | 0 |
| NH₃ | H | 1 | 0 | 2 | 0 |
| CO₂ | C | 4 | 0 | 8 | 0 |
| CO₂ | O | 6 | 4 | 4 | 0 |
This table shows how often stable molecules end up with zero formal charge.
How to get better at formal charge?
- It’s not about doing hundreds of problems. It’s about noticing patterns.
- After a while, you’ll look at a structure and already have a sense of where charges might be.
- That’s when it starts to feel less like a formula and more like intuition.
Final thoughts that feel a bit more real
Formal charge isn’t the most exciting part of chemistry, but it quietly supports a lot of understanding. It helps explain why molecules look the way they do and why some structures just don’t work. At first, it feels like counting. Then it turns into something else more like checking whether a structure “makes sense. And that shift doesn’t happen instantly. It builds slowly, one calculation at a time.